Eicosanoids
Eicosanoids (from greek eikosi − twenty) are compounds that derive from polyenic unsaturated fats with a 20 carbons long chain. They include prostanoids and leukotriens. In the prostanoids group (sometimes wrongly called prostaglandins) are included prostaglandins, prostacyclines and thromboxans. Generally the function of eicosanoids is to provide signal transduction (they interact with receptors connected to G-proteins). The mechanism of action is paracrine or autocrine. They influence the contraction and relaxation of smooth muscle tissue, blood coagulation, pain or for example inflammation. Eicosanoid's half life is extraordinary short, a few minutes.

Prostaglandins[edit | edit source]
Prostaglandins were discovered in seminal plasma. They are present in almost every tissue (kidneys, myocard, lungs, arterial wall and elsewhere), and act there as local hormones. Their synthesis consists of cyclisasing in the middle a twenty carbons long unsaturated fatty acid chain (e.g. arachidonic acid) resulting into a cyclopentane ring.
Three different eicosanoic acids (eicosapentenoic acid ω3, 20:5; arachidonic acid ω6, 20:4; dihomo-γ-linoleic acid ω6, 20:3) bring to light three different classes of eicosanoids characterised by the number of double bonds in the collateral chains (e.g. PG1, PG2, PG3). Differences in substituents determine a variety of types in every prostaglandin or thromboxane class (see below), marked with letters A,B, etc. (e.g. PGE1, PGE2 atd.).
Thromboxans[edit | edit source]
Thromboxans, discovered in thrombocytes (thanleukocytes and mast cells), contain a six-membered oxygen heterocycle (pyran ring).
Leukotriens[edit | edit source]
Leukotriens were firstly described in leucocytes. Their synthesis is influenced by lipooxygenase - it doesn't come to the chain cyclisation of fatty acids. What characterise them is the presence of three conjunct double bonds.
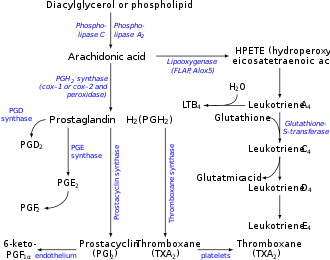
Synthesis of eicosanoids[edit | edit source]
For the eicosanoid's synthesis is essential the arachidonic acid (eicosatetraenoic acid) present in membranes as phosphotidylinositol. From here is lysed by phospholipase. During the treatment of an infectious disease (corticosteroids), the enzyme is inhibited, thus it results in a lower ARA production.
Synthesis via the eicosanoid pathway (cyklooxygenase, glykooxygenase path, cyclic path)[edit | edit source]
With this process are synthesised prostaglandins, prostacyclins and thromboxans. The source is the arachidonic acid, in some cases two 18C unsaturated essential acids (linoleic acid and α-linoelic acid), which can transform themselves in the needed twenty carbon long compound. Eicosanoids develop from ω6, and ω3 polyenic acids.
The name cyclic pathway derives from the fact that everything leads to the creation of a cycle in the middle of a 20C long chain. This gives us the derivatives of prostanoic acid (trans-7-(2-oktyl-1-cyklopentyl)heptane acid). The first step is the cyclisation of arachidonic acid actuated by prostaglandinendoperoxidsynthase (cyklooxygenase). Cyklooxygenase is one of the "suicidal" enzymes, because is capable, with its autokatalytic destruction, of stopping another sequential synthesis. The reaction runs aerobicly with the creation of endoperoxide. After slicing the peroxide bridge, with the help of other enzymes, it brings to light prostaglandins and prostacyclins.
During the creation of tromboxans, that incorporate an oxane ring, the endoperoxidase's structure is translated by the enzyme thromboxansynthase. During the synthesis of prostacyclines is used the prostacyclinesynthase resulting in prostacyclin I2. From it are derived other prostacyclins.
Cyclic paths that transform arachnoid acid (ARA) can be stopped with acetylsalicylic acid (Acylpyrin).
Synthesis by the lipooxygenase path[edit | edit source]
The lipooxygenase path creates hydroperoxyeicosatetraenolic acids (5-HPETE), which are linear and than lipoxins. With this path are synthesised other odourants e.g. form linoleic acid cis-3-hexenale with a typical smell of pruned meadows.
ARA is oxygenated during catalysis by 5-lipoxygenase. The hydroperoxide substitution is possible in various places of the molecule.
Main products of the path are active leukotriens, synthesised by various: HPETE → LTA4 → LTC4 → LTD4 → LTE4 a LTB4. Leukotreins with index 3 develop from eicosatrienoic acid and leukotriens with index 5 from eicosapentaenoic acid.
Lipoxins are antagonists LTC4, that have an antiflogistic (anti infectious) function.
Function of eicosanoids[edit | edit source]
As mentioned earlier, while talking about prostaglandins, the function is mainly about influencing the contraction of smooth muscles (blood vessels, intestine, stomach, bronchi...). The impact of prostaglandines (type E and F) in the intestine influences the excretion of water and electrolytes and in the end causes diarrhoea. They influence also kidney's tubules, blood pressure, change the kidney's perfusion, evoke the secretion of renin, angiotensin and ADH. PGD2 evoke sleepiness, on the other hand PGE2 has influence on awakening. A lots of E prostaglandins evoke the relaxation of GIT muscular tissue and the muscle contraction of the uterus. Prostaglandins inhibit the secretion of HCl in the stomach (it was used even by old Chinese that gave to patients dry seminal plasma), that stimulates lipolysis and influences many other functions in our bodies.
Prostacyclins have a similar function as prostaglandins. They act on HCl secretion in the stomach, PGI2 inhibits the aggregation of thrombocytes (anticoagulant effect).
Thromboxans work against PGI2 during the aggregation of thrombocytes; heighten the arterial blood pressure and co-work in the course of infectious pathways.
Most of leukotriens have anti-infectious effect, chemotactically attract neutrofils and can support the production of reactive oxygen forms. They are at the base of anaphylactic reactions.
All eicosanoids are quickly inactivated, usually by hydrolysis.
Pharmacotherapeutic use[edit | edit source]
Pharmacotherapeutic use is almost a diametral opposite to the large scale of action in which eicosanoids take part in the human body. PGE1 is used during lower limb's vessel diseases, many PGE and their synthetic analogous are used against gastric ulcers, they influence the microcirculation in the gastric mucosa. PG are indicated by gynaecologists and obstetricians for an enhancement of the uterine wall tonus and for the induction of labor (PG changes the characteristics of the uterine collagen with the help of collagenases), for abortion or to regulate the menstrual cycle. A large use is offered also in Angiology or during the medication of high blood pressure.
Links[edit | edit source]
Related articles[edit | edit source]
External links[edit | edit source]
References[edit | edit source]
- KOOLMAN, Jan – RÖHM, Klaus-Heinrich. Color Atlas of Biochemistry. 1. edition. Prague : Grada, 2012. ISBN 978-80-247-2977-0.
- MURRAY, Robert K, et al. Harper's Biochemistry. 23. edition. Prague : H & H, 2002. 872 pp. ISBN 80-7319-013-3.
- LEDVINA, Miroslav, et al. Biochemistry for medical students. Part I. 2. edition. Prague : Karolinum, 2009. 269 pp. ISBN 978-80-246-1416-8.