Antitumor therapy
From WikiLectures
Biochemical principles of antitumor treatment
Antitumor Treatment Modalities[edit | edit source]
Local treatment:
- surgery
- radiotherapy
Systemic therapy:
Criteria for the choice of modality and type of drug:
- guidelines (international - NCCN, national - blue book, constitutional, etc.)
- specific situation (condition and age of the patient, comorbidities, mobility, profession, etc.)
- economic aspects (centralization of care for patients treated with expensive drugs, etc.)
Chemotherapy[edit | edit source]
- development after World War I, when nitrogen mustard mustard (alkylating agent) was used for the first time
- by interfering with the cell cycle tumor cells prevent their further division
- the most sensitive are rapidly proliferating cells and cells that have a reduced ability to repair DNA errors
- acts non-specifically, which leads to the characteristic undesired effects of the treatment (effect on physiologically rapidly dividing cells):
- temporary suppression of blood formation (hematopoietic cells of the bone marrow)
- GIT problems (mucosal cells of the alimentary canal)
- alopecia (hair follicle cells) and others
Division by mechanism of action[edit | edit source]
Mitosis inhibitors[edit | edit source]
- Vinca alkaloids ("mitotic poisons") – Vinblastine, Vincristine, Vinorelbine
- alkaloids of periwinkle, synthetically produced in use today
- they bind to the β-subunit of tubulin and thus disrupt the dynamics of growth and degradation of microtubules - there is no polymerization of microtubules (they depolymerize directly at increased concentrations)
- Indications: breast cancer, lung and others
- Taxanes - Docetaxel, Paclitaxel
- chemically diterpenes
- originally from Pacific yew (paclitaxel), now produced synthetically
- by binding to the β-unit of polymerized tubulin, they increase the affinity of tubulin units to each other - stabilization of the microtubules of the dividing spindle - stopping of mitosis during the transition from metaphase to anaphase
- indication: breast cancer, ovarian cancer, prostate cancer etc.
Substances interfering with DNA replication[edit | edit source]
- DNA precursors
- Antifolates - prevent the normal function of folic acid in the body
- Methotrexate – competitively and irreversibly inhibits DHFR (dihydrofolate reductase) – binds 1000 times more easily, part of many therapeutic regimens
- Pemetrexed – structurally similar to folic acid, in addition to DHFR it also inhibits thymidylate synthase and glycinamide ribonucleotide formyltransferase
- Purine analogs
- Pentostatin inhibits adenosine deaminase
- thiopurines inhibit the synthesis and metabolism of purines (Mercaptopurine)
- Pyrimidine analogs
- inhibit thymidylate synthase (5-FU, Capecitabine) – cancers of the GIT, breast etc.
- inhibit DNA-polymerase
- inhibit ribonucleotide reductases (Gemcitabine) – pancreatic cancer
- inhibit DNA methylation
- Ribonucleotide reductase inhibitors
Topoisomerase Inhibitors'
- Topoisomerase I inhibitors
- topotecan – ovarian cancer + SCLC
- irinotecan – colon cancer
- Topoisomerase II inhibitors
- etoposide - lung cancer, testicular tumors and others
- Topoisomerase II inhibitors with intercalation activity
- anthracyclines' = anthracycline ATB
- produced by strains of bacteria Streptomyces
- in addition to inhibiting topoisomerase II, it also acts as an intercalator (they are inserted between two DNA strands)
- Doxorubicin, Epirubicin – cancer of the breast, ovaries, hematological malignancies
Substances acting by an alkylating or intercalating mechanism'
- Drugs acting through an alkylating mechanism
- alkylating agents: transferring an alkyl group (CnH2n+1) to N7 of the imidazole ring guanineu
- cyclophosphamide – hematological malignancies
- Platinum cytostatics
- do not alkylate in the true sense of the word - they do not have an alkyl group - only a similar effect to alkylating agents
- bind with DNA to form intercalation bonds that prevent replication and repair processes
- CDDP (cisplatin), oxaliplatin, CBDCA (carboplatin) – the basis of combined chemotherapy regimens for many solid tumors (sarcomas, ovarian cancer, lung cancer)
- Non-classical alkylating agents
- Dacarbazine – malignant melanoma, hematological malignancies
- Temozolomide – glioblastoma G IV
- Alkylating and intercalating substances
- Bleomycin – glycopeptide ATB produced by ``streptomycetes
- indication: HD, testicular tumors
- Mitomycin – a "streptomycete" product
- breast cancer, bladder cancer
Enzyme inhibitors[edit | edit source]
- Inhibitors of farnesyl transferase – Tipifarnib
- prevents attachment of the Ras protein to the cell membrane
- when farnesyltransferase is inhibited, the Ras protein (K and N) can also be modified by geranylgeranyltransferase
- blocking of both pathways leads to strong toxicity of the preparation, making its use impossible
- in the clinical research phase
- Inhibitors of cyclin-dependent kinases (CDKi) – Seliciclib
- preferentially inhibits CDK2, 7 and 9
- in vitro activates apoptosis of malignant cells
- in the phase of clinical trials in the indication NSCLC and in leukemias
- proteasome inhibitors – Bortezomib
- proteasome inhibitor (inhibits its chymotrypsin-like proteolytic activity)
- leads to cell cycle arrest by stabilizing negative cell cycle regulators (proapoptotic proteins are not degraded, leading to induction of apoptosis)
- proven efficacy in Multiple Myeloma and Mantle Cell Lymphoma
PARP inhibitors (Poly ADP Ribose Polymerase inhibitors)
- PARP, together with the product of the BRCA 1/2 genes, is involved in the repair of 1 and 2 DNA strand breaks
- more effective in tumors with an inactivating mutation in the BRCA 1/2 gene
- Olaparib – promising results in hereditary breast cancer, ovarian cancer and prostate cancer
- Uncategorized
- Trabectidine
- isolated from catfish
- efficacy demonstrated for soft tissue sarcomas
- mechanism of action not fully clarified (apparently reduces molecular O2 to form superoxide by an auto-redox process near DNA, which leads to irreversible damage)
- Tensirolimus
- a specific mTOR (mammalian Target Of Rapamycin) kinase inhibitor that modifies pro-growth signals
- with excessive activation of mTOR, there is an increase in the concentration of cyclin D and HIF, which leads to the stimulation of VEGF production
- in renal carcinoma, where mTOR often has increased activity
- Oblimersen
- blc2 antisense oligonucleotide - blocks the production of the BCL2 protein - an inhibitor of apoptosis
- in the clinical research phase
Antitumor immunotherapy[edit | edit source]
Trying to stimulate the immune system to recognize and destroy tumor cells:
- by administering systemic cytokines
-
- cytostatic to cytolytic effect
- changes in surface molecules lead to an increase in immunogenicity
- indication: generalized kidney cancer, in hemato-oncology
- has an activating effect on T-lymphocytes
- indication: kidney cancer, malignant melanoma
- administering an attenuated strain of BCG (Bacillus Calmette-Guérin) in bladder cancer - reduces the risk of disease recurrence after resection
- adoptive immunotherapy - eg administration of donor lymphocytes - in the clinical research phase
- monoclonal antibodies' - see biological therapy
Antitumor hormone therapy[edit | edit source]
- ancient times, middle ages' - observation: castrates had almost no prostate cancers
- 1896 Beatson was the first to perform ovariectomy for breast cancer to stop the progression of the disease, leading to regression of metastatic involvement of the chest wall
- the oldest "biological" in the sense of targeted therapy
- mostly used for malignancies derived from hormone-dependent tissue
- in general, manipulation of the endocrine system can do:
- by exogenous administration of hormone'
- by administering a substance that 'inhibits the production or activity of endogenous hormones
- 'surgical removal of endocrine organs (ovariectomy, adnexectomy)
Inhibition of hormone synthesis[edit | edit source]
- Gonadotropin Releasing Hormone (GnRH)
- stimulates the production of LH and FSH in the body
- administration leads to chemical castration
- after a certain period of administration (depot form) the increased production of LH and FSH leads to the down-regulation of receptors for LH and FSH in the ovaries or testes, which results in a decrease in testosterone in men and estrogens in women to the castration (menopausal) level
- before the onset of the effect, there will paradoxically be an increase in secretion - the need to administer a receptor antagonist
- goserelin – breast and prostate cancer
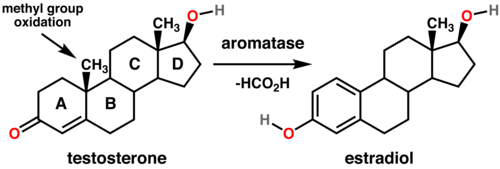
- Aromatase inhibitors (AIs)
- aromatase (AR) is an enzyme responsible for a key step in estrogen biosynthesis – it aromatizes androgens to form estrogens
- AIs competitively and reversibly inhibit ARs
- are used in postmenopausal women with receptor-positive breast cancers
- letrozole, anastrozole
Hormone receptor antagonists[edit | edit source]
- Selective Estrogen Receptor Modulators (SERMs)
- acts on the estrogen receptor
- different activity in different tissues - agonistic effects in some tissues - depends on coactivators and conformation of estrogen receptor
- tamoxifen
- both antagonist and agonist (e.g. on the endometrial mucosa – risk of hyperplasia up to endometrial cancer)
- indicated for hormone-positive breast cancers in pre- and postmenopausal patients
- biologically active only after activation in the liver parenchyma by the enzyme CYP2D6 (various isoforms, some so-called "bad metabolizers" - tamoxifen is then not effective enough)
- fulvestrant
- only estrogen receptor (ER) antagonist, down-regulates and leads directly to ER degradation
- in postmenopausal ER+ ca breasts with tamoxifen failure
- Antiandrogens
- androgen receptor antagonists
- often in combination with GnRH analogues or with surgical castration – so-called total androgen blockade
- prostate cancer treatment
- flutamide
- competes with testosterone and DHT for binding to the androgen receptor
- bicalutamide
- replaced flutamide for less intensity of side effects
- binds to the androgen receptor and accelerates its degradation
Others[edit | edit source]
- some hormone receptor agonists can have an antiproliferative or cytotoxic effect
- Progestogens – megestrol
- the principle is not fully clarified
- a direct effect on tumor cells and an indirect endocrine effect are assumed
- III. line of hormone therapy ca mammy, endometrium and prostate
Androgens
- previously at ca mom's
- Estrogens - diethylstilbestrol
- suppression of testosterone production
- in prostate cancer
- the mechanism is not fully understood - they probably reduce the incorporation of uridine into RNA and thereby the effectiveness of RNA polymerase, which ultimately leads to a reduction in RNA and protein synthesis
- part of chemotherapy regimens or in monotherapy for hemato-oncological malignancies
- CLL, multiple myeloma, lymphomas
- prednisone, dexamethasone
- Somatostatin analogs
- synthetic analogues of the peptide hormone somatostatin.
- somatostatin inhibits the activity of some adenohypophysis hormones (GH, FSH) and the production of GIT peptide hormones (gastrinu, motilin u, VIP, GIP etc.), thereby reducing GIT secretion and motility
- used for biologically active neuroendocrine tumors – VIPoma, gastrinoma, insulinoma
- indicated in carcinoids with carcinoid syndrome
- radioactive octreotide is also used in octreoscan
- octreotide (Sandostatin)
Biological treatment (targeted therapy)[edit | edit source]
- blocks the growth of tumor cells by affecting specific molecules important in the process of carcinogenesis, metastasis and cell growth (difference: classical chemotherapy "attacks" all rapidly dividing cells)
- usually the whole spectrum of rather non-specific side effects of X chemotherapy
Monoclonal antibodies ("-mab")[edit | edit source]
- Monoclonal antibodies against tyrosine kinase receptors
- Cetuximab (Erbitux)
- chimeric (mouse/human) monoclonal antibody (IgG1) against EGFR
- in EGFR expressing, KRAS wildtype (unmutated generalized colorectal carcinomas; mCRC) and in head and neck carcinoma
- Trastuzumab (Herceptin)
-
- downregulates HER2/neu, which cannot dimerize and thus does not initiate the PI3/Akt and MAPK signaling pathway (P27Kip1 is not phosphorylated, enters the nucleus and can inhibit cdk2 activity)
- inhibits angiogenesis
- "marks" tumor cells for the immune system
- in breast cancer with her2/neu overexpression
- in the Czech Republic, overexpression must be proven both by immunohistochemistry (IHC +++) and by fluorescence in situ hybridization (FISH)
- the main adverse effect is cardiotoxicity
-
- Monoclonal antibodies against other structures in solid tumors
- Bevacizumab (Avastin)
- humanized monoclonal antibody against VEGF
- the first clinically used angiogenesis inhibitor
- in combination with chemotherapy in mCRC
- clinical studies for other diagnoses are ongoing even without generalization
- side effects from inhibition of angiogenesis: hypertension - risk of CMP, kidney damage
- Catumaxomab
- binds EpCAM (epithelial cell adhesion molecule) to tumor cells with one arm and with the other T-lymphocyte and another immunocompetent cell with an Fc fragment - triggers an immune reaction
- used in the therapy of malignant ascites
- Monoclonal antibodies against other structures in leukemias and lymphomas
- Rituximab (MabThera)
- chimeric monoclonal antibody against CD20 protein found on maturing B-lymphocytes (no longer on plasma cells)
- mechanism of action not entirely clear (probably a combination of several additive mechanisms)
- treatment of B-lymphomas, leukemias and some autoimmune diseases
- Alemtuzumab
- antibody to CD52 is found on mature lymphocytes but not on stem cells
- second-line therapy in B-CLL, T-lymphomas
- Gemtuzumab
- antibody against CD33, expressed on most leukemic blasts
- in AML
Small molecule inhibitors of kinases ("-inib")[edit | edit source]
- inhibit specifically one or more protein kinases
- can be categorized according to the AMK whose phosphorylation they inhibit
- most often tyrosine kinase inhibitors
- mostly "small molecules" → penetrate biological barriers well X Ig
- Inhibitors of the receptor tyrosine kinase family - ERB (EGFR)
- HER1/EGFR'
- Erlotinib (Tarceva)
- binds reversibly to the binding site for ATP – prevents autophosphorylation and thus signal initiation
- indication: NSCLC (non-small cell lung cancer) after failure of at least 1 line of CHT
- with gemcitabine in generalized pancreatic cancer
- Gefitinib
- similar to Erlotinib; indicated in NSCLC
- HER2/neu
- Lapatinib (Tyverb)
- is a dual inhibitor – it binds to the binding site for ATP receptor tyrosine kinases in both EGFR and Her2/neu and thus prevents autophosphorylation and signal initiation
- able to act against so-called tumor stem cells (cancer stem cells, CSC) – they have the properties of normal stem cells – e.g. produce all types of cells in the tumor, it is assumed that they are responsible for relapses and metastases
- indicated for the therapy of Her2/neu overexpressed breast Ca
- Neratinib
- Inhibitors of receptor tyrosine kinases of class III
- Sunitinib (Sutent)
- inhibits several receptor tyrosine kinases (PDGFR, VEGFR, c KIT (CD117), RET etc.)
- indicated in metastatic renal cell carcinoma and in imitanib-resistant gastrointestinal stromal ttumors (GIST )
- Sorafenib (Nexavar)
- inhibits several receptor tyrosine kinases
- is unique in blocking the Raf/Mek/Erk (MAP-kinase) signaling pathway
- in advanced or metastatic renal cancer and hepatocellular carcinoma
- Inhibitors of receptor tyrosine kinases - VEGFR
- Vandetanib - in clinical trials for SCLC
- Semaxanib - in the phase of clinical trials for CRC
- Cediranib - in the phase of clinical trials for RCC, SCLC
- Axitinib' - in the phase of clinical trials for RCC
- Sunitinib
- Sorafenib
- Toceranib - used for therapy mastocytomas
- Regorafenib
- Inhibitors of non-receptor tyrosine kinases
- Imatinib (Glivec)
- in GIST, CML and dermatofibrosarcoma protuberans
- CML with t(9;22) - Philadelphia chromosome - translocation produces a fusion protein bcr-abl, which is a constantly active tyrosine kinase whose activity is reduced by imatinib, but also binds to c-kit and PDGFR
- binds to the ATP binding site
Links[edit | edit source]
References[edit | edit source]
- National Comprehensive Cancer Network. National Comprehensive Cancer Network: Your Best resource in the Fight against Cancer [online]. ©2009. [cit. 2009-12-20]. <https://www.nccn.org/default.aspx>.
- Czech Oncology Society ČLS JEP. Principles of cytostatic treatment of malignant oncological diseases [online]. ©2009. [cit. 2009]. <https://www.nccn.org/default.aspx>.
- Czech Oncology Society ČLS JEP. Chemotherapy or cytostatic treatment [online]. [cit. 2009]. <https://www.linkos.cz/pacienti/lecba/f_chemo.php/>.
- Czech Oncology Society ČLS JEP. Antitumor hormonal treatment [online]. [cit. 2009]. <https://www.linkos.cz/pacienti/lecba/f_hormon.php/>.
- Czech Oncology Society ČLS JEP. Targeted biological therapy [online]. [cit. 2009]. <https://www.linkos.cz/pacienti/lecba/f_bio2.php/>.
- Czech Oncology Society ČLS JEP. Targeted biological therapy [online]. [cit. 2009]. <https://www.linkos.cz/pacienti/lecba/f_bio2.php/>.
- Czech Oncology Society ČLS JEP. AISLP Drug Database [online]. [cit. 2009]. <https://www.linkos.cz/lekar-a-multidisciplinaryni-tym/ekonomika/sukl-databaze-leciv/>.
- State Institute for Drug Control. Medicines database SÚKL [online]. [cit. 2009]. <http://www.olecich.cz/leky>.
- NLM. TOXNET (Toxicology data network) [online]. [cit. 2009]. <https://toxnet.nlm.nih.gov/>.