Immune system development
The immune system is a set of mechanisms that maintain the integrity of the body. It is a complex system of cells and molecules that have the ability to recognize and eliminate foreign and own potentially harmful structures. Immunocompetent cells arise from a common stem cell in the bone marrow and then mature and differentiate in central (primary) lymphatic organs (bone marrow, thymus, ..). Immune reactions take place in peripheral (secondary) lymphatic organs, which are either anatomically defined structures (lymph nodes, spleen) or functional units (mucosal and cutaneous immune system, the immune system of individual organs). Disorders of the structure or function of the immune system can cause an increased susceptibility to infections, the development of autoimmune diseases, allergies or tumors.[1]
Structure of the immune system[edit | edit source]
| Innate immunity
(also non-antigenically specific, congenital, non-adaptive) |
cellular | phagocytes |
| macrophages | ||
| NK-cells | ||
| humoral | complement | |
| interferos (IFN) | ||
| Specific immunity
(also acquired, adaptive) |
cellular | T-lymphocytes |
| humoral | B-lymphocytes → antibodies (Ig) |
- Professional phagocytes: polymorphonuclear leukocytes (neutrophils) and monocyte-macrophages;
- Macrophages – arise from monocytes by entering the tissues, in different tissues they have different forms and different names (lymph nodes: histiocytes, skin and mucous membranes: Langerhans cells, CNS: microglia, liver: Kupffer cells, kidneys: intraglomerular mesangial cells, bones: osteoclasts,…);
- NK-cells (natural killers) – cytotoxic lymphocytes that are able to quickly kill virus-infected cells and some tumor cells.
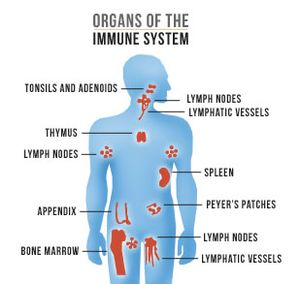
Immune system organs[edit | edit source]
- Thymus, bone marrow, lymph nodes, spleen, nasal and cervical tonsils, Peyer's patches in the intestine, appendix.
Cells of the immune system[edit | edit source]
- Lymphoid linage: T-lymphocytes (mediate a specific immune response), B-lymphocytes (provide specific antibody immunity), NK cells (cytotoxic cells of nonspecific immunity)
- Myeloid linage: monocytes – macrophages (antigen-presenting phagocytic cells), dendritic cells, neutrophils (phagocytic cells of the early inflammatory response), basophils (peripheral blood cells involved in the inflammatory response), eosinophils (cells involved in the hypersensitivity reaction and antiparasitic defense), mast cells (type I hypersensitivity reaction cells)[2]
Development of the immune system[edit | edit source]
The development of the immune system begins before birth and continues throughout life. Cells of the immune system develop from hematopoietic stem cells that are capable of unrestricted mitotic division.
In the 3rd week of gestation a pluripotent hematopoietic stem cell is formed in the yolk sac, which in the 5th week of gestation travels to the fetal liver (1st hematopoietic organ of the embryo) and temporarily also to the spleen. From the liver in 8-11. During the week of gestation, the stem cells are sown by embryonic circulation into the bone marrow, thymus, spleen and lymph nodes. After childbirth the only hematopoietic organ is the bone marrow.
The newborn has a functioning immune system (it is capable of both humoral and cellular immune responses) and the first months of life is additionally protected by transplacentally transmitted IgGantibodies from the mother. Their transmission occurs from the 22nd gestational week due to specific Fc-receptors in the placenta. The breastfed newborn is further protected by the mother's IgA obtained through breast milk, however, these IgA protect against pathogens in the digestive tract and do not enter the newborn's bloodstream.
The lowest level of immunoglobulins in the infant's serum is around 4 to 6 months of age, because passively acquired maternal IgG have already broken down by this period and endogenous immunoglobulins synthesis is not yet sufficient. It is during this period that the clinical manifestation of humoral immunodeficiencies occurs.[3][4]
T-cell development[edit | edit source]
The thymus is populated with fetal liver T-cell precursors ("pro-T cells"). TCRs ( T-cell receptors ) are developing and, thanks to the random recombination of genes, an enormous diversity of TCRs. TCR expression is followed by selection:
- Positive selection– interaction of immature thymocytes with low TCR expression with the main histocompatibility complex (MHC, genetic system responsible for self-recognition from foreign - especially HLA complex, Human Leucocyte Antigen ) on the epithelium (CD4 - HLA II, CD8 - HLA I) → cell selection capable of interacting with a foreign antigen (Ag) presented by its own MHC.
- Negative selection – thymocytes with high TCR expression reacting with own peptides presented with HLA I or II on thymic macrophages → induction of apoptosis → deletion of autoreactive clones.
The T cells then migrate to the secondary lymphatic organs.[3]
B-cell development[edit | edit source]
The development of B-lymphocytes takes place in several stages:
- Ag-free phases (antigen-independent development):
- In the bone marrow, it develops from progenitor cells: HLA-DR+, CD45+, CD34+, CD19+.
- It rerequires contact with bone marrow stromal cells (VCAM-1 + early lymphocyte VLA-4), cytokine SCF (Stem Cell Factor) and IL-7.
- Naive (virgin) mature B-lymphocytes have IgM, IgD on the surface.
- In the bone marrow, there is a negative selection of autoreactive clones of B-lymphocytes (those that strongly react with their own Ag) - induction of apoptosis, induction of rearrangement of gene segments for BCR, blockage and anergy.
- Phase with Ag stimulation (antigen-dependent development):
- In secondary lymphoid organs (node, spleen, mucosa), B-lymphocytes come into contact with T-lymphocytes and antigen-presenting cells.
- It takes place in 2 phases - primary and secondary phases of the antibody response:
- Primary phase of the antibody response (primary response)
- It occurs in primary lymphoid follicles (nodules), where B-lymphocytes interact with Ag on APCs and with Th2.
- When a naive mature B cell comes into contact with an antigen (Ag), the following occurs:
- To recognize antigen (Ag) via antigen-specific receptors on B-lymphocytes (BCR = B-cell receptor);
- To stimulate B cells by BCR + Ag binding;
- To absorb Ag → presentation of Ag at HLA II. cl. Th-cell precursors → formation of antigen-specific Th2.
- B-lymphocyte= APC forT-lymphocyte.
- Contact of T-lymphocytes + B-lymphocytes is enabled by CD28 + CD80 / 86, CD40L + CD40 and leads to the multiplication of B-lymphocytes and differentiation into:
- Secondary phase of the antibody response (secondary response)
- After repeated exposure of the memory B-cell to the antigen, a larger number of cells is produced and is accompanied by
- Afiinity maturation = BCR change;
- The of secondary lymph follicles with a germinal center;
- Isotype switching IgG, IgA, IgE;
- Formation of plasma and memory cells with higher affinity for Ag (somatic mutations of Ig genes increase the affinity of antibodies).
- Result of secondary response = Ig with higher affinity for Ag, able to activate complement and opsonize (IgG + FcR).[3]
Both primary and secondary responses immediately follow each other during typical infections and lead to the formation of memory cells. In case of recurrent infection (later encounter with Ag):
- Ig levels persist - immediate suppression of infection.
- Memory cells are rapidly activated to produce high affinity Ig of various isotypes.[5]
- Terminal maturation to plasma cells:
- Differentiation on memory bb. (for a certain Ag) and plasma cells (secretion of Ag-specific Ig = antibodies).
- Ig isotypes: M, G, A, D, E.[3]
Postnatal lymphopoiesis[edit | edit source]
T -lymphocytes[edit | edit source]
There is a higher number of T-lymphocytes (CD3 +) in umbilical cord blood than in infants. The CD4: CD8 ratio is higher. T cells have the ability to respond to a mitogenic stimulus and elicit an antigen-specific response (e.g. BCG vaccine).
B -lymphocytes[edit | edit source]
There is a higher number of B-lymphocytes in the umbilical cord blood. However, they do not yet form the full spectrum of immunoglobulins (Ig). After stimulation with antigens (Ag) of the external environment, IgM are first formed (this ability is also present in immature B-lymphocytes). Total Ig levels are lowest around 3 to 4 months of age. The ability to produce Ig against protein Ag is present from birth, but the ability to produce Ig against polysaccharide Ag up to 2 years (a polysaccharide vaccine is not suitable, but a conjugate vaccine such as Haemophilus influenzae type b). The newborn is more susceptible to G- infection because the lack of IgM (= opsonins) causes imperfect phagocytosis of polymorphonuclear. Maternal IgG have the function of opsonins for most G+bacteria, specific IgGs against common viruses are sufficient. Premature babies have fewer maternal IgGs and therefore lower opsonization activity for all types of microorganisms.[3]
Development of lymphatic organs[edit | edit source]
- Thymus – at birth it has 2/3 of its adult weight, the largest is just before puberty, then there is a gradual involution.
- Peripheral lymphatic tissues – adult size up to 6 years of age, larger in the prepubertal period, then involution.
- Spleen – gradually grows into adulthood.
- Peyer's patches – gradually grow, the largest during adolescence.[3]
Links[edit | edit source]
Related articles[edit | edit source]
- Primary immunodeficiency • Defects in cellular immunity • Defects of humoral immunity • Severe combined immunodeficiency
References[edit | edit source]
- ↑ LEBL, J – JANDA, J – POHUNEK, P, et al. Klinická pediatrie. 1. edition. Galén, 2012. pp. 223. ISBN 978-80-7262-772-1.
- ↑ http://fvl.vfu.cz/export/sekce_ustavy/mikrobiologie/imunologie/Prednasky/02_8_bunky_organy_bariery.pdf
- ↑ Jump up to: a b c d e f g http://www.vfn.cz/pracoviste/kliniky-a-oddeleni/klinika-detskeho-a-dorostoveho-lekarstvi/
- ↑ http://jeeves.mmg.uci.edu/immunology/CoreNotes/Chap17.pdf
- ↑ Jump up to: a b eamos.pf.jcu.cz/amos/kpk/externi/kpk_2816/8.ppt