Autoimmune diseases
Autoimmunity (AI) refers to the body's immune response to its components - autoantigens. A potential autoantigen is any protein, a variety of carbohydrates and lipids. The immune response to such an antigen is both humoral and cell-mediated, the presence of autoantibodies and autoreactive T-lymphocytes can be demonstrated, which damage the body's tissues, thus developing autoimmune diseases. They mainly affect young women.
- Division of autoimmune diseases
- Localized (organ-specific) - the immune response is directed against a specific tissue or organ.
- Systemic (non-organ-specific) - affects more organs.
- Physiological mechanisms of auto-tolerance
- Clonal deletions - during the maturation of immunocompetent T- and B-lymphocytes, those that would react with the body's own antigens are eliminated.
- Clonal anergy - loss or reduction of reactivity of immunocompetent cells specific against their own antigens.
- Peripheral inhibition - suppression of a reactive immunocompetent cell by other regulatory cells of the immune system (especially TS - CD-8+ suppressors).
- Immunological privilege - e.g. cornea, testicular canals, myelin sheaths - creates a barrier that prevents the passage of T-lymphocytes or cells of these tissues express Fas-ligand on their surface, which binds to the Fas-receptor of activated T-lymphocyte and induces its apoptosis.
- Mechanisms disrupting self-tolerance
- Alteration of self-antigen by binding of another molecule.
- Cross-reactivity of the antibody against two antigens, one of which is intrinsic and the other foreign.
- Polyclonal activation of B-lymphocytes.
- The imbalance between TH and TS.
- Disclosure of sequestered self-antigen against which immunotolerance has not developed.
- Genetic predisposition (association with HLA).
Disorder of peripheral lymphocyte tolerance[edit | edit source]
T-lymphocytes[edit | edit source]
- increased expression of costimulatory molecules in the tissue: transgenic expression - B7, IL-2, naturally e.g. infection,
- disorders of expression of inactivating costimulatory pathway molecules on lymphocytes: CTLA-4,
- mutations leading to apoptotic signal disorder: Fas (lpr / lpr), Fas-L (gld / gld), IL-2-/-, IL-2Rα-/-, IL-2Rβ-/-,
- T cell-mediated suppression disorder: transfer of T cell subpopulations,
- polyclonal stimulation: superantigen.
B-lymphocytes[edit | edit source]
- polyclonal stimulation: LPS.
- A - Genetic factors
- MHC genes:
- MHC II: Type I DM.
- HLA DR4 (5×–6× > risk).
- HLA DR3/DR4 (25× > risk).
- MHC I: Ankylosing spondylarthritis HLA-B27 (90×–100× > risk)
- MHC II: Type I DM.
- Other genes:
- IL-2 gene polymorphism (DM type 1),
- gene near CTLA-4 (DM type 1),
- C2 and C4 genetic deficiency (SLE),
- Fas a FasL (ALPS).
- B - The role of infection in the autoimmune process
- Viral and bacterial infections can theoretically affect the onset and exacerbation of the autoimmune disease.
- Infectious microorganisms are not present in the autoimmune lesion, which means the cause of autoimmune damage is not the infection, but the result of an immune response that can be induced or dysregulated by the presence of the microorganism.
- Potential mechanisms of influencing the autoimmunity of infections,
- costimulator expression (B7, IL-2, IFN-γ,…),
- alterations of own antigens, which may thus become partially cross-reactive,
- access to "sequestered" antigen (intraocular proteins, spermatozoa),
- molecular mimicry = cross-reactivity against foreign and self-antigen.
- C - Other factors in the development of AI disease
- Anatomical tissue damage (inflammation, trauma, ischemia = access to "sequestered" antigen).
- Hormonal effects.
- Higher incidence in women than in men (SLE ~ 10F/1M; SLE model (NZW/NZB) F1 only in females and affected by androgen therapy).
Localized autoimmune diseases[edit | edit source]
- Autoimmune hemolytic anaemia (against erythrocytes), thrombocytopenia (against platelets), and agranulocytosis (against granulocytes).
- Diabetes mellitus type I (against B-cells of Langerhans islets).
- Chronic gastritis type A (against parietal cells of the gastric mucosa and intrinsic factor).
- Goodpasture's syndrome (against basement membranes, especially in the kidneys and lungs).
- Hashimoto's thyroiditis (against colloid and thyrocytes).
- Graves-Basedow goitre (against TSH-thyrocyte receptors) - toxic diffuse parenchymal goitre.
- Myasthenia gravis (against acetylcholine receptors on neuromuscular discs).
- Primary biliary cirrhosis (against cells of the interlobular bile ducts of the liver).
- Nonspecific intestinal inflammation (IBD).
Systemic autoimmune diseases[edit | edit source]
- Collagenosis (lupus erythematosus, scleroderma, dermatomyositis).
- Rheumatoid arthritis.
- Sjogren's syndrome.
- Reiter's syndrome.
Viscerocutaneous collagenosis[edit | edit source]
- Systemic autoimmune diseases. These are fibrinous interstitial inflammations.
- They have nothing to do with collagen disorders - the name collagenosis was previously led to the finding that they can be treated with ACTH and corticosteroids.
A. Systemic lupus erythematosus (SLE)[edit | edit source]
- Formation of several types of autoantibodies against various antigens (and the resulting diversity of disease symptoms): against nuclear antigens (ANF - Anti Nuclear Factors) such as ds-DNA, histones, RNA, against phospholipids, etc. - binding of antibodies to antigens leads to the formation of immunocomplexes, which are stored in various tissues and damage them (activation of complement, whose chemotactic components attract neutrophils, which release their lysosomal enzymes) - this is an immunopathological reaction type III.
- It is characterized by the presence of hematoxylin bodies in the interstitium of damaged tissues, it is a histological picture of LE cells - granulocytes with large basophilic inclusion, which is the remnant of the phagocytosed damaged nucleus.
- It mainly affects young women.
- 1. Acute and subacute lupus erythematosus
- Skin:
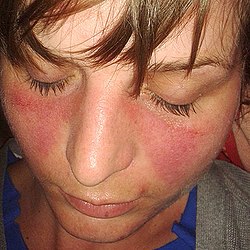
- Red-violet erythema + photosensitivity. Under the influence of UV radiation or other skin damage, autoantigens are released into the circulation, where autoantibodies bind to them and immunocomplexes are deposited in the basement membrane of the epidermis.
- Especially in places not covered by clothing (butterfly erythema on the face, palms and fingertips).
- Microscopically fibrinoid necrosis of the corium just below the epidermis, narrowing of the epidermis, vacuolar degeneration to the disintegration of the cells of the basal layer (subepidermal vesicles), thickened basement membrane.
- Vessels: Fibrinoid necrotic generalized vasculitis (analogy of PAN, but also affects smaller vessels).
- Kidneys: Focal glomerulonephritis.
- Heart: Libman-Sacks endocarditis (atypical verrucous endocarditis) - non-bacterial endocarditis, mainly affecting the ventricular area of clogged valves and the wall endocardium.
- Seroses: Non-purulent polyserositis (pleuritis, pericarditis).
- Joints: Arthritis, especially of the small joints of the hand.
- Blood: Disorders of hematopoiesis (cytopenia) and coagulation (thrombosis).
- 2. Chronic lupus erythematosus
- It is characterized mainly by skin involvement (foci with inflammatory erythema, follicular hyperkeratosis, scarring atrophy of the corium and periadnexal small cell infiltration), skin cancer is more common.
B. Scleroderma[edit | edit source]
- Sometimes it also affects internal organs (lungs - more common lung cancers, GIT - often oesophagus, muscle replacement with ligament - leads to stenosis and dysphagia, more common cancers, blood vessels - intimal thickening and circulatory disorders), has a limited and generalized form.
C. Dermatomyositis[edit | edit source]
- It affects the skin, muscles and nerves, in the acute stage, it resembles erythematosus, in the chronic it rather resembles scleroderma.
Rheumatoid arthritis (polyarthritis progressiva)[edit | edit source]
- Chronic disease of a large number of joints (usually symmetrical), conditioned by an immunopathological reaction type III (deposition of immunocomplexes - the so-called rheumatoid factor of IgM in binding with IgG antibodies – and immune responses to them - activate complement, whose chemotactic components attract polymorphonuclear cells – their enzymes cause disruption of the surrounding tissue), a common complication is secondary (AA) amyloidosis.
- It has some features in common with rheumatic arthritis (heart disease, findings of rheumatic nodules), but the pathogenesis is different, it more often affects middle-aged women.
- Clinical manifestations
- It begins as morning stiffness and soreness of the joints of the hand and foot, gradually decreasing the range of motion until ankyloses and muscle contractures develop.
- It is a permanently progressive disease that can lead to complete immobilization.
- Microscopy
- At each outbreak, the joint cavity is filled with serous to sero-fibrinous exudate of low viscosity, mixed with polynuclear cells.
- The synovial membrane is congested and permeated, with round cell cellulation (plasma cells form rheumatoid factor and immunoglobulins. The resulting immunocomplexes are deposited in the synovialis, cartilage and synovial fluid). The membrane is later swollen (villous productive synovialitis) and extends to the cartilage like a pannus. The cartilage necrotizes and granulation tissue forms beneath it (reminiscent of osteoarthritis, but there are no osteophytes at the edges of the joint).
- In the later stages, the synovialis smoothes out, the pannus turns into a thick fibrous membrane, fibrous and later bony ankylosis occurs, and osteoporosis develops around the joint, which later affects the entire bone.
- Macroscopy
- In the final stages, muscle contractures arise (mainly from limited joint movements due to pain), which leads to deformities of the limbs - the appearance of the hand is typical, where the contractures of the interosseous muscles create an ulnar deviation of the fingers.
Sjogren's syndrome[edit | edit source]
- The clinical syndrome, characterized by xerostomia, xerophthalmia and rheumatoid arthritis, takes the form of:
- primary - affects the exocrine glands (salivary, lacrimal, sweat, tracheobronchial, gastric, vaginal);
- secondary - in addition, manifestations of collagenosis (SLS, scleroderma, polyarteritis nodosa, chronic polyarthritis, Reynaud's syndrome), in 5–10% it causes MALTom.
- It typically affects women around menopause.
- The cause is probably an autoimmune process characterized by the production of antibodies against cytoplasmic antigens of the ductal epithelium. There is a possibility of an inductive role of sialotropic and lymphotropic viruses.
- Microscopically the same picture as myoepithelial sialoadenitis (lymphoid infiltrates around the outlets, which narrow, their lumen disappears and turn into epimyoepithelial islets, the relationship between the two syndromes is close, but in more than half of patients with myoepithelial sialoadenitis there is no xerostomia and/or xerophthalmia).
Reiter's syndrome[edit | edit source]
It consists of a trias:
- nongonococcal urethritis,
- post-infectious seronegative arthritis,
- conjunctivitis.
Current treatment of autoimmune and allergic diseases[edit | edit source]
- Anti-inflammatory drugs (NSAIDs, steroids);
- Symptom blockers (antihistamines, antagonists of leukotrienes and their receptors, soluble TNF-R…);
- Cyclosporine – immunosuppression;
- Plasmapheresis.
Experimental treatment of autoimmune and allergic diseases[edit | edit source]
- Induction of tolerance - repeated oral administration of antigen (ongoing clinical study of prevention of type I DM - insulin, therapy of rheumatoid arthritis - collagen type II):
- i.v. administration of antigen;
- repeated parenteral administration of low doses of antigen (clinical study of the prevention of type I DM - insulin).
- Influence of costimulation (blockers of costimulation molecules).
- Proinflammatory cytokine antagonists (such as IL-1, TNF) - TNF-αR.
- Gene therapy.
- Specific influencing of the course at the molecular level - blockers of effector molecules and their receptors. Blockers of lymphocyte migration into tissues.
- Immunoablation and autotransplantation of CD34+ cells - (clinical study in patients in late stages of life-threatening autoimmune diseases, e.g. lupus erythematosus, multiple sclerosis, autoimmune hemolytic anaemia, autoimmunne thrombocytopenic purpura etc.).
- Copolymer 1 (Copaxone), a myelin analogue composed of - L-tyrosine, L-glutamic acid, L-alanine, L-lysine (multiple sclerosis).
Allergy[edit | edit source]
Links[edit | edit source]
Related Articles[edit | edit source]
- Autoimmune liver diseases
- Systemic lupus erythematosus
- Systemic scleroderma
- Rheumatoid arthritis
- Reactive arthritis
References[edit | edit source]
- ŠTEFÁNEK, Jiří. Medicína, nemoci, studium na 1. LF UK [online]. [cit. 2010-02-11]. <https://www.stefajir.cz/>.
- PASTOR, Jan. Langenbeck's medical web page [online]. ©2006. [cit. 2011-10-27]. <https://langenbeck.webs.com/>.
| This article is a stub. You can join the authors and edit it. You can discuss the changes at discussion. |